Africa and Asia are the continents that have been hit hardest by the problem of counterfeit goods. It has always been so for a long time, but not for long.
Why?
It’s simple, these continents are fighting back!
How are they fighting back?
With strategic PARTNERSHIPS!
Over the years, we have seen several partnerships between progressive governments and innovative technology companies to tackle counterfeiting head-on, no holding back.
Afghanistan is one of such progressive government, while Fantom Foundation and Chekkit Technologies are two of such innovative technology companies.
The problem they set out to solve?
A lack of visibility in the pharmaceutical industry. Too many players (many of which are illicit) selling drugs (many of them fakes) that were harming everyday Afghans.
To put that in perspective, as of 2015, a total of 450 foreign pharmaceutical suppliers were registered with the health ministry in Afghanistan, which had a population of 31 million people. In comparison, India, which had a population of more than 1.2 billion, had about 100 registered foreign medicine suppliers. Clearly, something needed to be done to better sieve out the illicit pharma traders.
In that vein, the Afghanistan Ministry of Public Health has signed an MoU with Fantom and Chekkit to help tackle the problem of counterfeit meds in the country using blockchain. The project begins with a 3-month pilot and will utilize Chekkit’s product authentication technology to track and verify all drugs sold in the country.
The Fantom Blockchain Challenge (held in November 2019 at the AfricArena Summit in South Africa), in which Chekkit was among the top three participants, initiated the partnership. Fantom was already in conversation with the Afghan Ministry of Health and brought Chekkit into the discussion because our solution was advanced enough to tackle the Afghan counterfeit problem and the fact that Chekkit’s solution was built to work perfectly in developing countries, was a huge plus as well. The partnership sees Chekkit’s solution deployed on Fantom’s Opera blockchain network.
How Does The Drug Verification & Tracking Work?
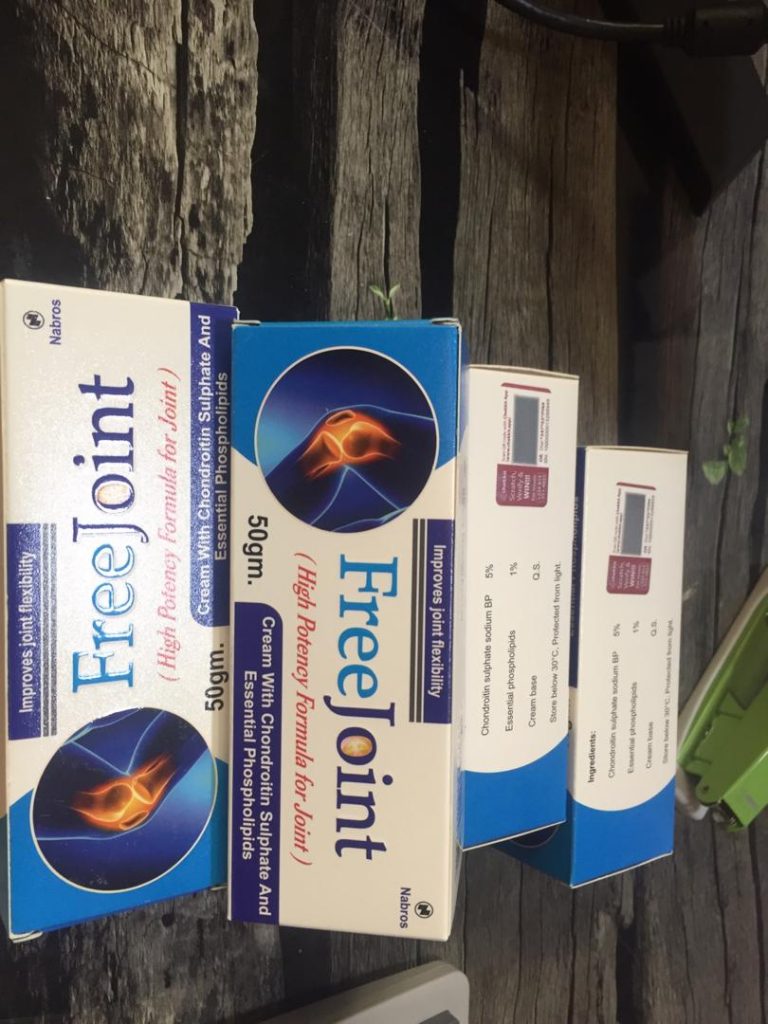
Chekkit’s smart labels will be attached to 80,000 pharmaceutical products sold in the Afghan market. These pharmaceutical products include;
- 50,000 items of hand sanitizer produced by Bliss GVS, endorsed by the World Health Organization, and recently launched in order to fight against Covid-19 in Afghanistan. Royal Star is the distributor of this product in Afghanistan.
- 10,000 items of free joint cream produced by Nabros Pharma. This cream help relieves joint pain.
- 10,000 items of Kofanol Chewable Tablets produced by Nabros Pharma. This tablet helps relieve cough, cold, and sore throat.
- 10,000 items of Diacare Foot Cream (Diabetic) produced by Nabros Pharma which is used in treating diabetic-related skin infections.
Each smart label has a unique QR code generated by Chekkit’s proprietary software which contains information on each product like its authenticity, manufacturing date, expiry date, FDA number, manufacturer, distributor, and so on.
On the part of the Afghan public/consumers, they can then verify the drugs before purchase/use by scratching Chekkit’s smart label on any of the pilot products to reveal the product’s QR code, scan with Chekkit App, and then view the authenticity of the product as well as other product information.
To encourage verification, Chekkit has deployed a loyalty/reward system where consumers win a little something back when they verify products. This is a win/win for everyone involved.
Chekkit has also provided an oversight capability for the Health Ministry by providing special hand-held devices that they can use to verify the authenticity of the products at the point of entry into the country. This is possible because our smart labels are made using special materials called taggants, providing an extra layer of security for all parties involved.
How Big is The Whole Project?
Speaking on the innovative initiative, Dare Odumade (our able Founder and CEO at Chekkit Technologies) said:
“The Afghanistan Ministry of public health had been looking for effective ways to verify the authenticity and effectiveness of drugs that are being imported into the country and Chekkit provided them with a way to authenticate the drugs at the point of entry into the country and also at point of purchase. We believe this double focus strategy will make it extremely difficult for counterfeiters to thrive.
Through this partnership, we will provide the pharma companies involved with valuable consumer insights and a reward program to encourage purchase and verification by buyers, as well as give the government/ministry a transparent view of the pharmaceutical market. On completion of a successful pilot, our technology will be deployed across the board for all meds coming into and produced in Afghanistan.
Finally, we will also deploy a supply chain tracking solution that offers 360-degree real-time track and trace protection against counterfeiting. The supply chain tracking will be implemented in collaboration with manufacturers, distributors, and retailers”.
Partners Involved

Chekkit Technologies: We are an African HealthTech startup providing a secure global product authentication system with practical applications across; anti-counterfeit protection, consumer data gathering & analysis, consumer rewards & loyalty programs, as well as personalized consumer engagement & communication.
Fantom: A DAG-based smart contract platform with fast confirmation times. Their solution is built on Fantom’s opera mainnet, providing fast and secure consensus capable of scaling to the high transactional throughput required to power a country’s entire healthcare system.
Afghanistan Ministry of Health: The Ministry of Health is committed to implementing these solutions throughout the healthcare system in Afghanistan and is interested in new and emerging technologies such as blockchain that will help solve various problems.
Royal Star Ltd: One of Afghanistan’s biggest distributors of pharmaceutical products. The company distributes products from ten producers located around the world such as the United Kingdom and India. They have also worked closely with bilateral and multilateral aid organizations such as the World Health Organization and USAID.
Nabros Pharma Pvt Ltd: A manufacturing and exporting company based in Ahmedabad, Gujarat. They are accredited by the World Health Organization Good Manufacturer Guidelines (GMP), the Pharmaceutical Inspection Co-operation Scheme (PIC/S) based in Geneva, Switzerland, and the National Agency for Food and Drug Administration and Control (NAFDAC), Nigeria. The company manufactures products such as tablets, capsules, small volume parenteral, ointments, and oral liquids which are exported to 35 countries around the globe.
Interested in learning more about chekkit’s anti-counterfeit and product verification software? Visit our website www.chekkitapp.com or just get a free demo now